Stoichiometry Guided Instructional Activities with Guide Framework
This set of three worksheets are intended to be used as collaborative "Guided Instructional Activities" (GIAs). Two students cooperate to complete the steps of a stoichiometry problem, alternately doing parts of the process as they explain what they are doing and evaluate their partner's work. These worksheets emphasize an algorothmic approach that helps students learn to think aobut the purpose of a question, organize their work, set it up so that it is easily readable and can be followed by others, and make good use of "unit analysis" (dimensional analysis). Good habits and organization are emphasized by introducing a "framework" in which students enter their work on the first two GIAs. Use of the framework is optional, and may be extended to other stoichiometry practice, or discontinued as soon as the teacher is satisfied that students are able to present their work neatly and successfully.
One worksheet is needed for each group of two students. Worksheets can be placed in sheet protectors and used from year to year.
These worksheets are part of a entire unit on teaching stoichiometry. You can access the complete lesson plans with information on their use, and links to other worksheets, labs, and activities at Stoichiometry is Easy.
Materials
One copy of GIA for each group of two. Copies may be placed in sheet protectors to be used over again year to year, or the GIA may be projected using a document or overhead projector.
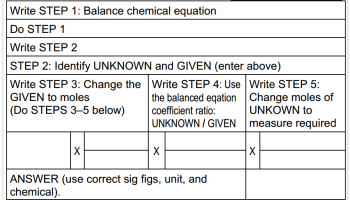
One "framework" for each group of two is also recommended, at least for the first two GIA activities. The frameworks are at this URL: /sites/www/files/StoichFrameworks.pdf
The first framework has the steps for stoichiometry written into it. The second framework in the file does not have steps, so students can write the steps into the framework for themselves as they begin to learn this algorithmic method.
Background
Most students, and many of my (now former) colleagues, find stoichiometry to be one of the most challenging topics in a first year (and yes, even a second year) chemistry class. But my students and I have always looked forward to the challenge, and the fun. Certainly, stoichiometry means diligent work, and for some it means frustration. But there is no reason it has to be dire, difficult, or drudgery.
I had vacillated over the years between using an algorithmic method, and an inquiry-based approach to teaching stoichiometry. After six years trying both methods in alternating years, I decided my students got more out of the algorithmic approach, and that in the process they learned several lessons that made the rest of their chemistry work easier. The five steps my students learned are by no means unique, but the frameworks included seem to help students better organize their work, gain an understanding of who the mathematical steps of stoichiometry relate to the chemical representation (equation), and encourage students to be more successful.
Procedure
Guided Instructional Activities (GIAs) were part of the “Mastering Chemistry on the Web” (MCWeb) program, a part of the National Science Foundation Molecular Science Project (http://www.molsci.ucla.edu). Dr. Patrick Wegner (California State University, Fullerton) developed these POGIL-like (Process-Orineted Guided-Inquiry Learning) activities for use in preparatory and general chemistry classes. While some of the activities are, like POGIL real guided inquiry, many are simply cooperative learning activities that give students an opportunity to work toward a common goal while discussing and practicing skills of particular interest.
The usual procedure is to assign students randomly into groups of two (which change for each activity). They work at tables of two groups each. Students who have difficulty with an item are to consult their partner, then the other group at their table, and then may ask the teacher. Students should not move from group to group. The teacher circulates around the class making sure each group is on task, answering questions, and revealing the answers a little at a time so students can confirm they are correctly doing what they were asked to do. This put students in charge of their own learning, gave some the opportunity to “teach” others, and allows the teacher more time to work with students who needed extra attention during class time in a non-threatening environment. At the same time, students police each other to make certain everyone (OK, nearly everyone) is on task.
These GIAs are part of a unit on stoichiometry. Read the entire article and find the lesson plans at: INSERT URL
Questions
See GIA in the student document below.
Concepts
These three activity worksheets guide students to complete stoichiometry problems using an algorithmic approach that emphasizes "unit analysis" (dimensional analysis) and the use of the "stoichiometri ratio" (coefficients of the relevant chemical species in the balanced chemical equation.
Time Required
Each of the activity worksheets requires 40 to 55 minutes.
Preparation
Make one copy of the GIA worksheeet for each group of two students (or arrange to project using overhead or document projector). Copies of the GIA may be placed in sheet protectors for use from year to year.
If desired, make one copy of the "framework" for each group of two students. Using the framework with the steps written in is suggested when having student complete the first GIA. Students can use the totally blank framework (and be instructed to write in the steps for themselves) for the second GIA. Use of the framework is optional for the third GIA.
Attribution
The GIAs were written by Dr. Patrick Wegner, California State University, Fullerton, and revised by David Licata, chemistry/AP Chemistry, Pacifica High School, Garden Grove, CA (retired). The framworks were created by Mr. Licata.