A Quick and Accurate Determination of the Heat of Fusion of Water

This activity is intended for Chemistry Teachers who plan to discuss the energetics of phase changes. It follows a recent article, Simple, Quick, and Accurate Determination of the Heat of Vaporization of Water, on the accurate determination of the ΔHvap of water at 100°C, using an electric kettle, which forms a necessary foundation for deep understanding of the concept presented here.
Materials
See the Supporting Information for access to the teacher resource. Readers must log into their account to access. Not a member? Register for free!
Background
Procedure
The following provides an accurate, empirically based1 determination of the ΔHfusion of water at 0°C. For students to get full value from this activity, I recommend:
- Students complete the above mentioned ΔHvap of water (@ 100°C) determination first, followed by a fulsome class discussion. The final question concerns the heating curve for water; understanding this is essential to the activity presented here. Students must also understand the how and why of the determination of the effective power output (wattage) of an electric kettle or hot plate.
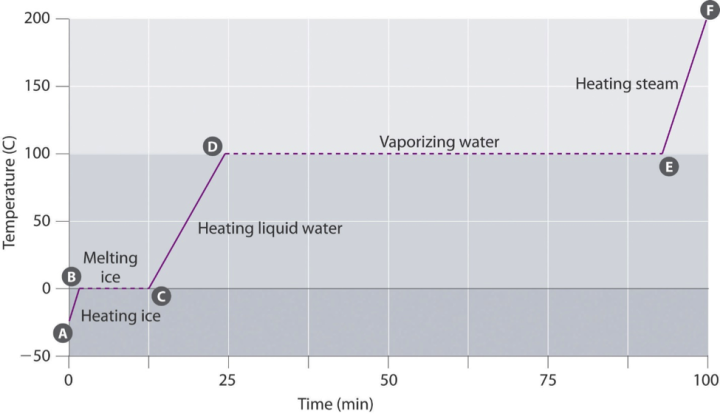
Figure 1: The Heating Curve for Water2 (See also an activity for determining a heating curve for water: Will a Watched Pot Ever Boil?)
- Students should complete the preparatory questions, given in the supplementary materials—and go over the answers in class—before proceeding to the activity.
Before I go on, let me say that I had a lot of trouble with the accurate determination of the ΔHfusion of water. I scoured old lab manuals—which I collect like baseball cards—to no avail. I simply could not make any of these procedures work to arrive at an accurate value for the ΔHfusion of water at 0°C. Here is what I came up with:
1. Begin by determining the effective power output of a small electric hotplate to heat—not boil—a beaker of water. A beaker that completely covers the surface of hotplate is recommended.
Table 1. Data Used to Determine the Effective Power Output (Wattage) of an Electric Hot Plate
| mass of water heated | 373.5 g |
| initial temperature of water | 29.8 °C |
| final temperature of water | 42.5 °C |
| heating time | 137 s |
| specific heat capacity of water3 | 4.18 J•g-1•°C-1 |
The wattage of the hotplate was calculated as follows:
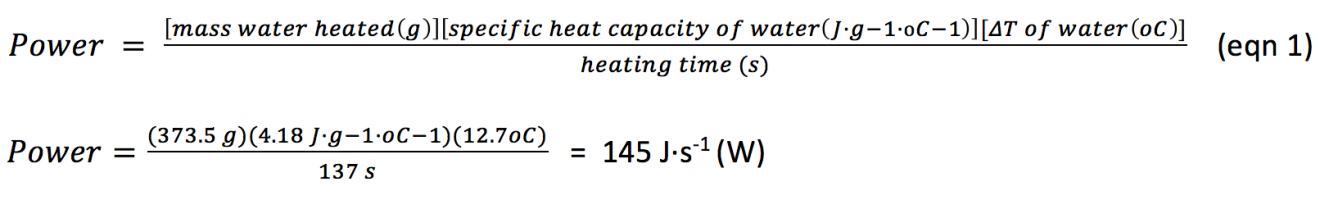
And so the electric hot plate has an effective power output of 145 W.4
2. The above apparatus was used to determine the heat of fusion of water. A measured mass of ice, taken from an equilibrium mixture of ice and water, which had been in an insulated container for about 15 minutes,5 was added to a known mass of water, whose initial temperature was measured. The beaker was immediately placed on the previously plugged-in—and warmed—hot plate; the timer was activated. With occasional stirring, the sample was heated to well beyond the point where the ice had melted. The final temperature and the heating time were recorded.
Table 2. Data for the Determination of the ΔHfusion of water at 0°C
| specific heat capacity of water6 | 4.18 J·g–1·oC–1 |
| effective power output of hotplate | 145 J•s-1 (W) |
| mass of water heated | 370.0 g |
| initial temperature of water, Tiwater | 5.5 oC |
| mass of ice added to water immediately before heating | 22.0 g |
| initial temperature of ice, Tiice | 0.0 oC |
| final temperature of water and melted ice, Tf | 30.0 oC |
| heating time | 331 s |
To calculate ΔHfusion of water at 0oC from the above data, we employ the equation:
Energy, E, supplied by hotplate = E to melt ice at 0.0 oC + E to heat melted ice from 0.0 oC to Tf + E to heat water from Tiwater to Tf (eqn 2)
Substituting data:
(145 J·s–1)(331 s) = (ΔHfusion, J·g–1)(22.0 g) + [(22.0 g)(4.18 J·g–1·oC–1)(30.0oC – 0oC)] + [(370.0 g)(4.18 J·g–1·oC–1)(30.0oC – 5.5oC)]
Solving for ΔHfusion gives
ΔHfusion of water at 0oC = 334 J·g–1 or [334 J·g–1][18.0 g·(mol H2O)–1] = 6.01 x 103 J·mol–1 or 6.01 kJ·mol–1
To two significant figures, this duplicates the literature value of 6.02 kJ·mol–1.7
In the interest of time, I have never had a class of students carry out this investigation. I use it as data analysis activity.8
Taken together with the empirical determination of the ΔHvap of water (at 100oC), this activity illustrates the straightforwardness—and accessibility—of these determinations. Students see that determination of these values is not “rocket surgery”.
See the Supporting Information for access to the teacher resource. Readers must log into their account to access. Not a member? Register for free!
Notes and References:
- Data obtained by the author in a High School Chemistry lab.
- 6.8: Heating Curve for Water - Chemistry LibreTexts
- It is assumed that the temperature of an equilibrium mixture of ice and water in an insulated container is 0oC
- The manufacturer’s indicated power of the hotplate was 330 W.
- It is assumed that the temperature of an equilibrium mixture of ice and water in an insulated container is 0oC.
- 17.4: Heat Capacity and Specific Heat - Chemistry LibreTexts
- Heat of Fusion and Vaporization (utexas.edu)
- That said, I urge you to have your students try this, even if it’s in your after-school Chemistry Education Research Group/Chemistry Club.
Questions
Concepts
heat of fusion
Time Required
Preparation
Distribute document and have students complete the preparatory questions before solving for the heat of fusion of water.
Attribution
N/A