Mole Conversions: 1- & 2-step with format
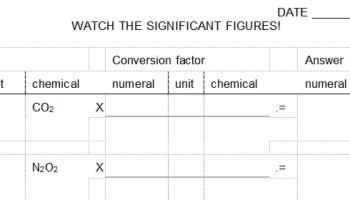
This worksheet asks students to do basic conversions of mass or molecules to moles and vice versa. The worksheet requires students to complete their work in a particular format and to inlcude number, unit, and chemical identity for each item in the "given," in each conversion factor, and in the answer. It gives students basic practice in this mathematical exercise while inforcing good habits that encourage "unit analysis" (or dimensional analysis). See below for a link to how this worksheet is used within an entire unit on the basic mathematics for introductory chemistry and stoichiometry. Answers are part of the student document. The teacher document contains blank format pages that may be used for additional practice.
This activity may also be used as a homework assignment.
This worksheet is used early in the course when teaching conversions of various units to and from moles. It is an essential component of lessons leading to stoichiometry. You can access the complete lesson plans with information on their use, and links to other worksheets, labs, and activities at https://www.chemedx.org/article/stoichiometry-easy.
If you are logged in to your ChemEd X account, you will see the worksheet available for download in the supporting information.
Materials
Photocopy of worksheet.
Background
Teachers will recognize the utility of "unit analysis" or "dimensional analysis" when students must carry out unit conversions. This worksheet enforces the good habits of requiring students to list numeral, unit, and chemical identity for each item in a mathematical problem. By determining which units and identities "cancel" students should be able to confirm when they have the mathematics correctly set up and thereby guarantee a correct answer (provided they can work a calculator). In reviewing the mathematical calculation, I believe it is always important to point out that when items "cancel" it means that students are dividing one quantity by the identical quantity, and therefore they end up with the mathematical identity: 1. Of course multiplying or dividing by 1 does not change the value in the answer.
Procedure
Students complete the worksheet.
Questions
See worksheet.
Concepts
Unit analysis (dimensional analysis), mole conversions
Time Required
This worksheet can be used as an in-class or as a homework assignment. The ten items on the first page should take 20 to 30 minutes. The ten items on the second page should take 30 to 50 minutes.
Preparation
Photocopy the worksheet.
Attribution
Created by David P. Licata, Chemistry & AP Chemistry, Pacifica High School (retired)