Lewis Dot, VSEPR Shape, Polarity and Intermolecular Forces Activity Sheet

Determination of Lewis Dot structures and visualization of the shapes of molecules using valence shell electron pair repulsion theory (VSEPR theory) is an example of an abstract concept that students often find difficult to learn. I have found it useful to have a single worksheet/packet that my students can add to as we cover Lewis dot structures, resonance, VSEPR shapes, polarity, and intermolecular forces.
These topics are typically covered in the “Chemical Bonding” and “Molecular Geometry” chapters of textbooks. Having one worksheet seems to allow students to connect one topic to another as we proceed with our lessons and build concepts. We save some class time by referring to columns already filled in instead of redrawing Lewis structures of molecules, for example. After completing this worksheet, I find that students are able to draw structures, assign shapes, describe the polarity and intermolecular forces present for almost any simple molecule or ion I give to them. The worksheet includes both molecules and ions and examples that follow and many that violate the octet rule. Students learn to identify the “best” Lewis dot structure by minimizing formal charge to create expanded octets. Students work in pairs and use the model kits seen in Figure 1. Figure 1 is a copy of the picture I give to students within the model kit that describes what each of the pieces represents. Along with these worksheets, students and I use the “Bear Essentials Of Polarity” comic book1 to explore polarity and a number of PhET simulations to look at molecular shapes2 and molecular polarity.3

Figure 1: Labeled Model Kits
Kinesthetic (tactile) learners seem to favor building molecules with the model kits while visual learners tend to state a preference for the PhET interactive simulations, but all learner styles seem to benefit by completing this worksheet. My students have improved their test scores by an average of 9.43% (±1.27, n = 495 post and n = 1230 pre-use) over these topics since I have used the single worksheet, molecular models and PhET simulations. The VSEPR Summary Sheet (found in Supporting Information) is given to students only after they have completed the PhET simulation “Molecule Shapes Interactive Simulation”.2 After practicing with the models and other tools, I have students work some of the examples for Lewis Structures without the use of model kits or simulation software so they can learn how to write them out by hand. They do not have access to the PhET simulations during tests (although I do allow them to use the molecular model kits if they choose). If you are logged into your ChemEd X account, you will see the student worksheet and the teacher’s answer key in the Supporting Information.
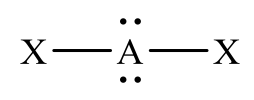
Figure 2: Example Lewis structure
Student misconceptions seem to derive from their lack of understanding of 2-dimensional representations of 3-dimensional structures. For example, students will often times see 4 bonds to a central atom in a central molecule and believe the bonds are 90° apart because they are thinking in 2-D (x,y) space. Using the model kits, students can see that the correct arrangement of those electron pairs is actually the 109.5° idealized angle. If a student (correctly) draws figure 2 as their Lewis structure, they will often assign the angle as 180° instead of <109.5.
Working with models helps them to visualize these situations in 3-D space more accurately. Another misconception that I frequently encounter occurs with octahedral molecules. Students hear the “octa-” prefix and assume 8 atoms are bonded to the central atom instead of 6. They are not experienced enough to understand that there are 8 faces to an octahedron, not 8 atoms.
Log into your ChemEd X account to download the Student and Teacher Documents.
Materials
“Minit Organic/Inorganic Set” (Item # 840230 from Carolina Biological Supply)
Tool Bench Hardware Storage Cases (purchased from my local Dollar Tree store)
Six Minit Organic/Inorganic kits were combined to make a total of 15 of the student kits I put together.
Background
I usually have an introductory discussion lesson in which I outline Lewis Dot symbolism. We review valence electrons, electronegativity, concepts of ionic and covalent bonding, and formal charge during this lesson. I present the steps involved in drawing Lewis structures:
- Sum the valence electrons (adding 1 for each negative (–) charge on an anion and subtracting 1 for each positive (+) charge on a cation)
- Draw the skeletal structure with a single bond to connect each atom to the central atom (with the most electropositive atom usually acting as the central atom)
- Add remaining electrons to complete the octet of those atoms needing additional electrons
- Shift unshared pairs of electrons into bonding positions if needed to satisfy the octet rule and/or minimize formal charge (which usually creates expanded octets)
Procedure
- I distribute the worksheet to students and we complete the valence electron count, Lewis structure, and formal charge columns together as a class for H2O, CBr4, and PI3. (10 minutes).
- Students work in pairs to complete the same columns for the next 5 examples (15 minutes). We review those answers together before proceeding.
- As time allows, we complete and review more examples in class. Students are asked to complete those columns for all examples for homework. We review their answers (and the correct answers) during the next class meeting (30-40 minutes).
- We discuss resonance and what makes one structure preferred compared to an alternative arrangement. Students complete the 3 columns with the correct answers and then turn their packets in.
- VSEPR Theory is introduced using the PhET “Molecule Shapes Interactive Simulation”2 This is a guided activity that introduces students to valence shell electronic pair repulsion theory (VSEPR). It is a computer based activity that takes approximately 60 minutes. Students work in pairs and complete the activity sheet.
- The original student worksheets are returned to students so that we can complete the electron pair geometry and molecular geometry columns of the packet.
- I pass out the model kits and give students time to examine the different shaped pieces. I also pass out the VSEPR Summary Sheet for their use. We discuss what each piece represents and I have them practice making models from the Lewis structures for the first 3 examples on their original worksheets (15 minutes).
- I then skip to HCN to show students how to make multiple bonds with the kits and how the molecular shape is determined by the relative positions of the nuclei.
- Students working in pairs are then asked to make models as needed to complete the electron pair geometry and molecular geometry columns of the packet (30 minutes).
- Answers are reviewed together and students can correct their answers as necessary (10 minutes).
- The student packets are again collected.
- I next have students complete the “Bear Essentials of Polarity” worksheet and comic book (30 minutes).
- We summarize what is meant by polarity and when a molecule will be polar or nonpolar.
- Together, the students and I work the PhET “Intermolecular Forces and Molecules - Interactive Lecture Demonstration” (45 minutes).
- We continue to discuss polarity and intermolecular forces present in molecules/ions.
- The student worksheets below are once again returned to students (with all but the final 2 columns completed). We work the first 5 or 6 examples together as a class.
- Students then work in pairs to complete the last 2 columns of the worksheet (30 minutes).
- We review the answers for all molecules and ions (10 minutes). Students correct their worksheets as needed. The sheets are now to be used as study guides for students prior to their unit assessment. All of the Lewis, VSEPR, polarity, and intermolecular force examples are collected in one place for their use.
Questions
Concepts
Time Required
Approximately 3 hours of classtime is required plus some homework time.
It may take about 3 hours to initially prepare the kits. This will need to be done only once, but the kits should be checked at least once per year to replace missing “atoms”.
Preparation
- The model kits were put together using the “Minit Organic/Inorganic Set” and Tool Bench Hardware Storage Cases. Six Minit Organic/Inorganic kits were combined to make a total of 15 of the student kits I put together. There are pieces left over to replace those that “get lost” over time. It took about 3 hours to initially prepare the kits. This will need to be done only once, but the kits should be checked at least once per year to replace missing “atoms”. Pictures of the kits with labels are part what I distribute to students so they become familiar with what each piece represents.
- A copy of the student worksheets found below, the Molecule Shapes - Guided-Inquiry Activity (by Timothy Herzog & Emily Moore) from the PhET website, and the Bear Essentials of Polarity Comic Book and worksheet should be printed for each student. (1 hour). The comic books can be reused from year to year.
- Computers are needed for the simulation activities. We are a BYOD school, but others may want to prepare by reserving a computer lab or a school computer cart for the PhET simulations.
Attribution
- Investigation IV – Molecules in Action Lesson 4 – Polar Bears and Penguins from Smells © UC Regents, LHS Living by Chemistry, 2003. I have found it online through a Google search for “Bear essentials of Polarity”. It is sometimes titled “Penguins and Polar Bears”.
- Molecule Shapes Interactive Simulation: http://phet.colorado.edu/en/simulation/molecule-shapes I use the Molecule Shapes - Guided-Inquiry Activity (by Timothy Herzog & Emily Moore) found under the Teacher’s on the main simulation page. https://phet.colorado.edu/en/contributions/view/3947 (accessed 5/21/19)
- Molecular Polarity Interactive Simulation: https://phet.colorado.edu/en/simulation/molecule-polarity Intermolecular Forces and Molecules - Interactive Lecture Demonstration https://phet.colorado.edu/en/contributions/view/3959 (accessed 5/21/19)