An Activity to Demonstrate the Principles of Chemical Kinetics and Equilibria
Over the last ten years I have taught general chemistry at several junior colleges, frequently to students who have limited background in chemistry and weak mathematic skills. Through experimentation, I have found a technique to actively engage a class in understanding chemical kinetics, chemical equilibria and appreciating why chemists introduced the concept of “the mole.” Inspiration for this goes back to my undergraduate mentor (G.G. Long at NC State Univ. in 1966) who, faced with a similar problem, described a dynamic equilibrium as two children tossing sand back-and-forth between opposite ends of a sand box. With some trepidation, I decided to put a modification of this analogy to the test, and found it works rather well when properly staged.
This is a hands-on student-participation demonstration, that (1) requires no toxic chemicals, requires no safety equipment and generates no waste; (2) is easy to set-up and clean up; (3) is suitable for students with minimal chemical knowledge; (4) provides students with easily visible demonstration of a dynamic equilibrium; (5) demonstrates the relationship between rates and equilibria in a semi-quantitative numerical format; (6) gives students a practical demonstration of why chemists work in moles; (7) give an explicit demonstration of the effect of concentration on rate of reaction, (8) it only takes about 30 minutes...and (8) the students seem to think it is a lot of fun.
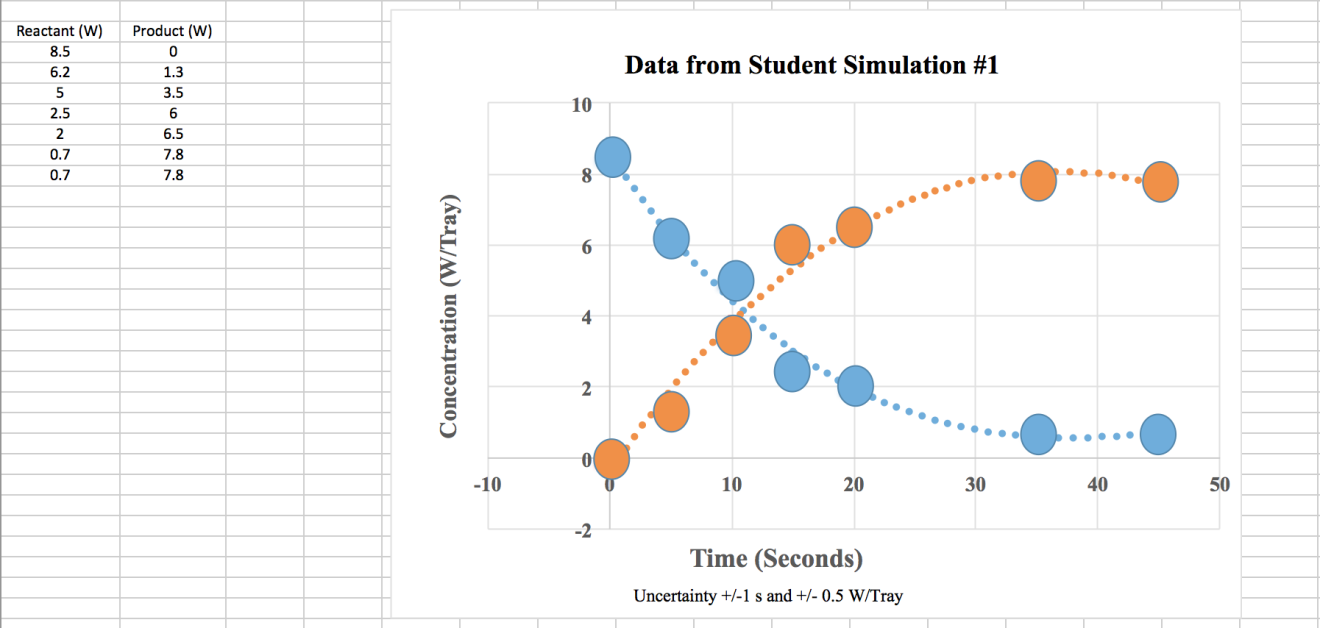
Figure 1 - Sample Student Data
Materials
two large (disposable) aluminum serving pans (30 cm x 50 cm)
two large bags (1.2 kg each) of small (M&M™) candies (2 g or less) representing at least 1000 pieces
two scoops of different sizes and shapes (e.g., a plastic laundry detergent scoop and a medium size paper cup). It is best if the two scoops are of similar size and they can be identical.
Background
I generally invoke this activity when introducing chemical kinetics and equilibria. At this point, students should know how “the mole” is defined but may not appreciate why it was introduced. They also typically have had lectures on chemical reactions, balanced equations and basic thermochemistry and enthalpy. Thus, I begin the lecture by making the point that it is not obvious from either the equation of the reaction or the heat of reaction how fast the reaction will proceed. Typically, I provide energy diagrams on the board showing reactants and products and explain that the rate of reaction depends – in part- on the height of the barrier between these two states (activation energy) while the direction of the reaction depends on the difference in free energy of the two states. Transition state theory assumes that reactants are in equilibrium (usually termed pseudo equilibrium) with transition state molecules (activated complex) and that the relative amounts of reactant and transition state is related to the height of the energy barrier (activation energy) and available thermal energy (RT) as related by the Arrhenius equation. Moreover, once in the transition state, the rate of forming products is limited only by the rate at which the chemical bonds can change (i.e., vibrate). I generally explain heat capacity during the lectures on thermochemistry in terms of storage of kinetic energy in translation, rotation and vibration of molecules. And, during discussions of light (electromagnetic radiation), I explain that the infrared part of the spectrum corresponds to the frequency of vibration of molecular bonds (~1014 s-1). At this point, I can present the Arrhenius equation (to students in the sciences and engineering) or go directly to the rate constant (k) if the class is not prepared for the Arrhenius concept.
In the simplest case of reaction
A D B
And, assuming first-order kinetics, I can write for the disappearance of A, in terms of the concentration of A = [A]
RateA = -kA [A]
Likewise for B
RateB = -kB [B]
And when and equilibrium is reached, the forward rate and reverse rate must be equal:
RateA = -kA [A] = -kB [B] = RateB
Which can be reduced to the equilibrium constant (Keq):
Keq = kA/kB = [B]/[A]
With this background the class is prepared to execute the activity.
Procedure
For this activity, I generally use two large (disposable) aluminum serving pans (30 cm x 50 cm); two large bags (1.2 kg each) of small (M&M™) candies (2 g or less) representing at least 1000 pieces; and two scoops of different sizes and shapes (e.g., a plastic laundry detergent scoop and a medium size paper cup). Overall cost of the materials is about $30.00, but some can be reused. I arrange the trays on opposite sides of a small table (with long edges in contact and taped together) and pour at 500 to 1000 candies into one of the trays (covering the bottom evenly about 1 layer thick). If the number of candy pieces is larger than this, the kinetics actually become zero-order; i.e. rates become independent of the concentration (candies per tray or candies per square-centimeter) and depend only on the size of the scoops (Keq = kA/kB but not the ratio [B]/[A]). I explain that the candies will represent molecules and that the two trays will represent “A” reactants and “B” products (in the equations above). The concentration of reactants or products is essentially measurable in “candies per tray.”
From the class, I ask for two volunteers whom I describe as “competitive and athletic but not aggressive.” These students come forth and are assigned the scoops. The one with the larger scoop stands behind the tray with the candies (reactant). Then, I ask for a student with a stopwatch (everyone with a cell phone has a stop watch) to volunteer as the timer. This student is positioned (sitting or standing) at the end of the table (between the trays). Another volunteer is selected to be a recorder and moves to the white board, on which I have derived the expression for Keq shown above and provide blank tables for recording data (as shown in the supplemental data).
Rates and Rate Constants
The actual rate of scooping up candies (from a nearly full tray) and passing them to the other tray is approximately zero order such that the rate equals the rate constant:
RateX ~ kX
By measuring the rate of transfer of candies from the reactant tray (A) to the product tray (B) for a short time (~5 to10 s), we determine the rate and rate constant for the forward reaction. A typical student with a medium-sized (300 mL) paper cup can move about 50% of the candies in 10 seconds. With a small scoop (100 mL), the student in the role of the reverse reaction can usually move about a third of the candies in 10 seconds. Any candies that are ejected from the pans should be returned to the pans.
But, how do we measure the candies? Counting them individually would be a time-consuming activity; and I point out to the students that this is exactly the problem that chemists have with molecules. Thus, I take an arbitrary container (e.g., 90 mL paper cup) and define it as one of the student’s name, e.g., “the Jones” …after all “the mole” has already been taken! In a few minutes, the candies have been measured in “Joneses” and the recorder calculates the rate in “Joneses/second” (one significant figures, e.g., 0.4 Joneses/s). This is typically repeated two or three times and averaged, or the median value is taken if the data are scattered. The students are usually slow in the first trial and this measurement can be deleted as an outlier.
If you like, you can go farther on this path by weighing the “Jones” so that the number of Joneses can be determined by weight. You can also have a student (e.g., Jones) count the number of molecules in a “Jones” to determine “Jones’s number” (analogues to Avogadro’s number). In one case, I had my students do a statistical analysis of the masses of individual candies (mean, and standard deviation), which can be likened to isotopes.>
Once the forward and reverse rates and rate constants have been determined, the equilibrium constant is predicted from the ratio of the forward and reverse rates.
The Equilibrium Constant
Now, the excitement begins as all the candies are placed in the reactant side and the two students (reactant and product) face off. On signal from the timer, reactant starts moving candies to the product side and product simultaneously begins returning them.
If time permits, the action can be stopped every 5 s and the number of candies in each tray are counted and the progress of the reaction can be tabulated and plotted, (i.e., “candies per tray”) as a function of time.
To save time, I allow the students to proceed for a full 30 seconds before counting the candies in each pan (in “Joneses” of course). Usually, within 30 s the analogue is near equilibrium. After recording these data, the reaction is resumed for another 30 s; and the pans are again counted. If necessary, the equilibrium is continued for a third period of 30 s; and the pans are counted again. In my experience, after a total of 90 seconds, an equilibrium has been established very close to the ratio predicted by the measured rates (rate constants). This seems to work because as the concentration of candies thins out, the system approaches first-order kinetics (i.e., the rates depend on the concentration of candies rather on the size of the scoop). The equilibrium constant is typically between 1 and 2, depending on the sizes of the scoops and motivation of the students.
Clean Up & Waste Disposal
Clean up is easy: All the candies passed back and forth and handled during measurements are discarded. The second bag of unused candies are then shared by the students, who at this point seem to have a cheerful understanding of rate and equilibrium constants and a greater appreciation of why chemists work in moles. Safety issue: In performing this experiment do not use any glassware for the project. Only paper and plastic cups and plastic or metal scoops.
Experience: Simulation of chemical equilibrium
I have done this demonstration approximately 20 times with students in introductory chemistry and environmental science classes at three different colleges. It takes between 30 and 45 minutes to complete the work. You can download the entire Teacher Document, including three recent examples and notes below.
Questions
Concepts
kinetics, equilibrium
Time Required
30 - 45 minutes of class time
Preparation
I arrange the trays on opposite sides of a small table (with long edges in contact and taped together) and pour at 500 to 1000 candies into one of the trays (covering the bottom evenly about 1 layer thick). I explain that the candies will represent molecules and that the two trays will represent “A” reactants and “B” products. The concentration of reactants or products is essentially measurable in “candies per tray.”
Attribution
A similar game-like activity can be found in the supporting information of this JCE Laboratory Excercise: Exploring Chemical Equilibrium with Poker Chips: A General Chemistry Laboratory Exercise, Thomas H. Bindel, Journal of Chemical Education 2012 89 (6), 759-762, DOI: 10.1021/ed200406t.
Another similar JCE Laboratory Excercise was just published ASAP as this ChemEd X was being reviewed for publication. Using Beads and Divided Containers To Study Kinetic and Equilibrium Isotope Effects in the Laboratory and in the Classroom, Dean J. Campbell, Emily R. Brewer, Keri A. Martinez, and Tamara J. Fitzjarrald, Journal of Chemical Education Article ASAP, DOI:10.1021/acs.jchemed.6b01004.
Credits
General
Items tagged with the safety:general tag have some safety concerns that may be also described more specifically by additional safety terms
General Safety
For Laboratory Work: Please refer to the ACS Guidelines for Chemical Laboratory Safety in Secondary Schools (2016).
For Demonstrations: Please refer to the ACS Division of Chemical Education Safety Guidelines for Chemical Demonstrations.
Other Safety resources
RAMP: Recognize hazards; Assess the risks of hazards; Minimize the risks of hazards; Prepare for emergencies