Acids & Bases Interactive Notebook
We’re coming close to the end of our Interactive Notebooking journey – this is my second to last unit – Acids and Bases. As a continuing theme from earlier, I am moving to focus more on particle view diagrams in combination with symbolic representations and macroscopic observations.
The flow of this unit has evolved over the years for me. When I first began using interactive notebooks, my acid-base unit began with properties of acids and bases and the different acid base theories. If this is information that is important to your class, I have included the foldable for this as bonus material. Instead, I now start with a comparison of strong versus weak acids. Out of that conversation comes a discussion of the concept of conjugates, but less of the focus is on classifying substances as Arrhenius versus Brønsted-Lowry. As I have mentioned before, I’ve been switching the placement of notebooks within my unit structure. This week I used the strong versus weak page as an exit ticket to check the comprehension after we had finished a strong versus weak PhET activity and a card sort. I used the PhET as the leftside of the notebook page. I had my students do a guiding Google Doc, and then a QR code can be pasted on the page directly to take them to the SIM. I use this SIM often during this unit, and having it as a permanent reference in their notebooks is very helpful. (Some of the particle view cut-and-paste images from the Strong versus Weak page are pulled from the PhET.)
I next get into the math of acids and bases. This year, I focused my instruction on whole number pH values, which was a GAME CHANGER and I will always introduce pH this way from now on. The page I have for the math relationships ties well into any level of math instruction. It is designed to highlight the physical relationship between the values of pH and the concentration values for H+ and OH-.
The output page for this spread could take many, many forms. A task card activity with a tracker would fit well here, as would a traditional worksheet. As I write this post, I’ve been inspired to create a cut-and-paste ranking activity. This is new and hasn’t been used in my classroom, but I’m excited about the relationships it can help students synthesize. I hope you like it!
Finally, I have a spread on titrations. Some years I get to this page, and some years I don’t. This year we did a titration in a semi-quantitative way with a focus on the species present instead of titration calculations, so I didn’t use this page.
The output side of the titration spread is titration data graphing and analysis. This has students return to graphing skills, and would nicely complement a lab in which students use a probe that may generate a graph through a graphing program. There is also an excellent virtual titration experiment from the Royal Society of Chemistry that would be a great digital output option if you can’t do a full titration in your class.
Materials
Composition notebooks, printed copies of each page, colored paper or construction paper for making flipbooks/foldables, scissors, tape or glue
**A note on my manipulatives: If you see a dashed line, that is where the item should be cut. If you see dotted lines, that means fold.
Background
For backstory about how Nora uses interactive notebooks, view a recording of her ChemEd X Talk: Integrating Interactive Notebooks into Chemistry Courses with Nora Walsh and read her previous post, Interactive Notebook Unit on Stoichiometry. Nora wrote the following in response to questions she received after her ChemEd X Talk: A Classroom View of Using Interactive Notebook Pages.
See all of the interactive notebook units Nora has published: INTERACTIVE NOTEBOOK COLLECTION
Procedure
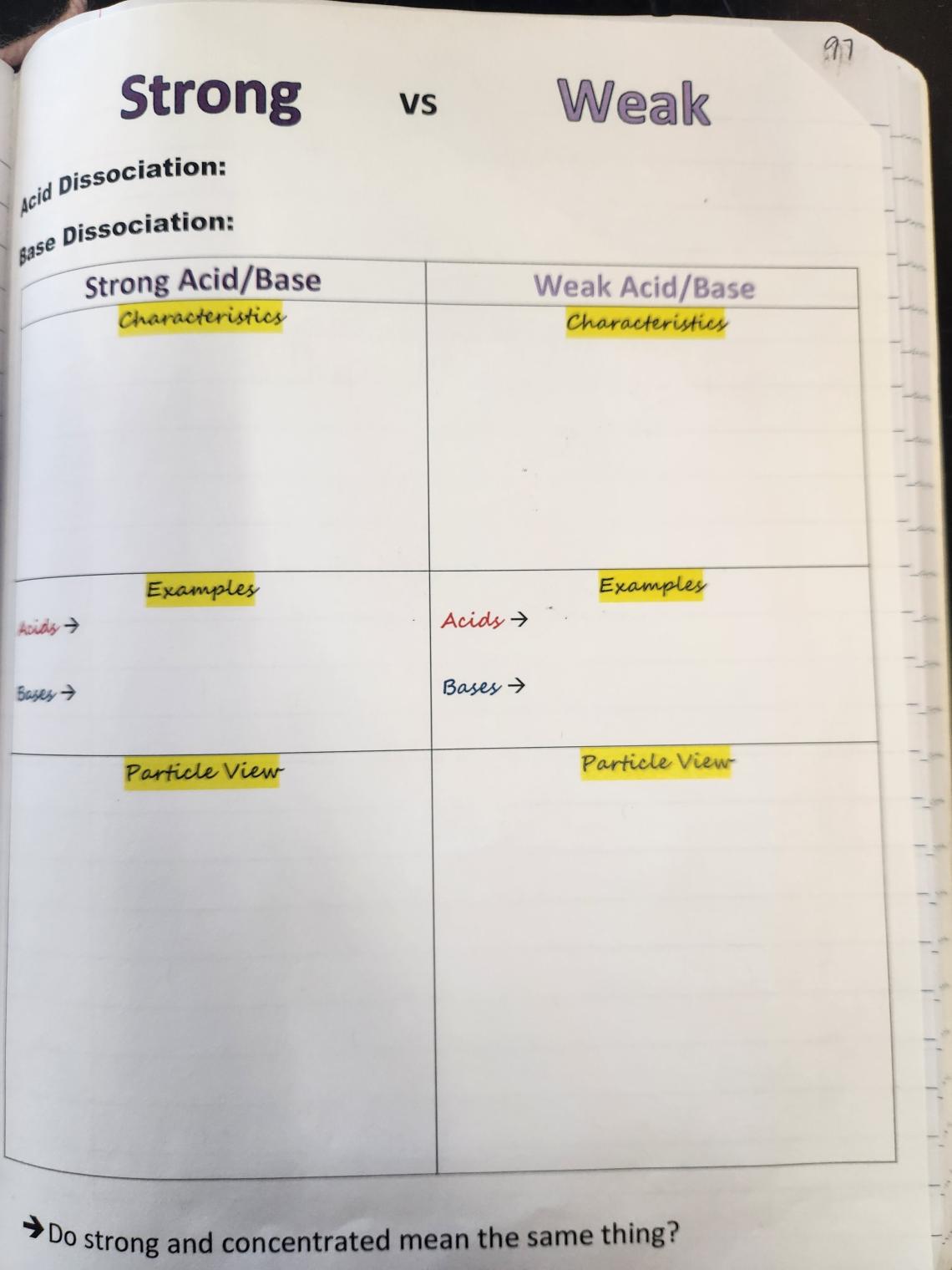
Spread 1: Strong vs. Weak Acids
Rightside: This is a primarily a page glue down that students fill in at the top with examples of acid and base dissociation equations, followed by a side-by-side comparison of strong versus weak characteristics and some common examples. Students fill in information with a focus on complete versus partial dissociation, and the bottom of the page has space for particle view representation cut-and-paste that illustrate examples of acids and bases in both strong and weak forms. The last item asks them a question to drill into a misconception: do strong and concentrated mean the same thing?
Leftside: As mentioned above, I have students work through the PhET simulation on Acid-Base Solutions. If you’ve followed all the way through the interactive notebook, you have seen me use PhET frequently, and this is one that I revisit multiple times throughout this unit. I would encourage you to also have your students use the QR code as a glue in, and then visit phet.colorado.edu/en/simulations/acid-base-solutions, log in to access the Activities page and then pick your favorite guiding document for your students’ exploration of this amazing resource.
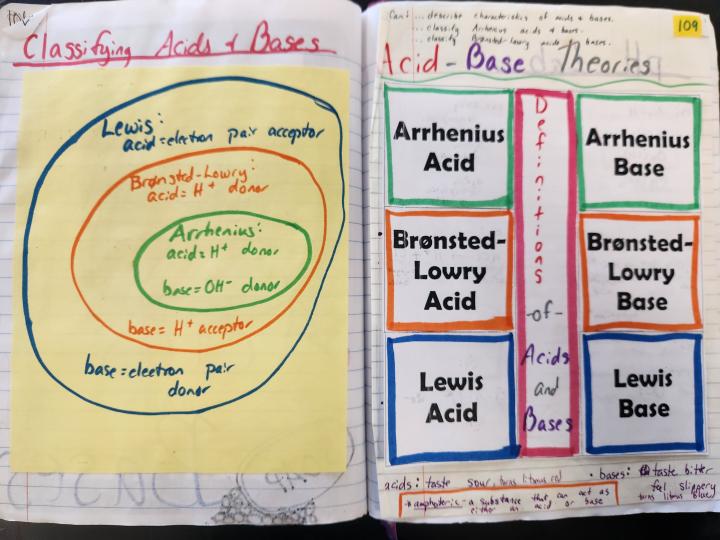
Bonus material: If you want to include the acid-base theories in your notebook, check out the bonus material foldable for this! I have students include the definition/feature of each theory and then reactions with labels that demonstrate the theory in action.

Spread 2: pH Calculations
Rightside: This is also a one page glue down to introduce the equations. The bottom is my favorite part of this page – it is a shutter foldable that gives a LOT of information all at once. Students can see the colors of universal indicator/pH paper and litmus, and it also shows real-world examples of each with the pH and pOH relationship showed. Opening the pOH flap lines up the concentration of H+ to the corresponding pH to drive home the meaning of pH. The opposite relationship can be seen using the other matching sets. You can use this foldable to really drive home the idea of a log is an exponent.
*A note on printing: If at all possible, I highly recommend you print the pH foldable in color. Additionally, if you need to limit your printing, the pH/pOH barndoor foldable is formatted to match the line spacing in college ruled composition books. There are two copies of the pH foldable per page, and students can line up the foldable to the lines on their page, eliminating the glue down, and you can have them fill in notes on equations directly on the notebook page.
Leftside: Newly created pH cut and paste ranking! Print the page for all students. Students glue the color coded arrow down the side of the page, and then cut out the solution descriptions. They then line them up from most acidic to most basic. This activity is done with whole pH values to really emphasize the relationships between the various ways of expressing acidity and basicity quantitatively.

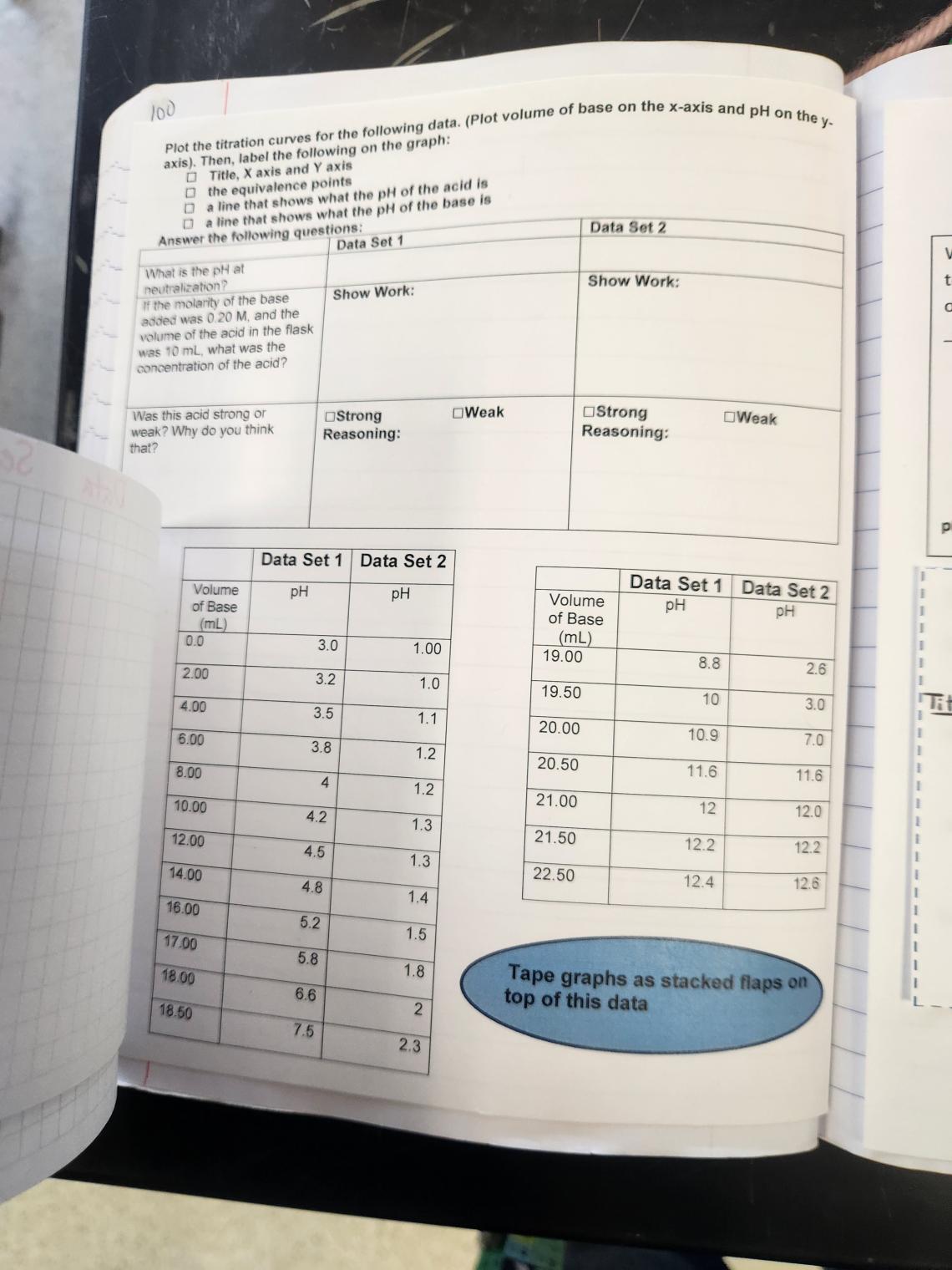
Spread 3: Titrations
Rightside: There is a lot of technical information on this page. I don’t often spend an entire spread on a lab procedure (maybe I should do that more?) but titration is a pretty fundamental and specific lab skill. The top left corner showing neutralization is to tie the student’s use of titration to a prior concept, and then the rest of the page takes students through the lab-specific vocabulary, indicator selection, general procedure, and equivalence point calculations at the bottom. The titration setup image is a flap to cover the vocabulary of titrations block, and the indicators and step images cover their complementary blocks. The MAVA = MBVB is a flap to just remind them of this previously-seen formula.
Leftside: This output has students plot titration data for two sets of data, and then compare the curves to determine if they show the titration of a strong or weak acid with justification. The graphs are taped as flaps on top of the data tables for direct comparison.
It may be helpful to view video 1 to help visualize what these pages will look like in action.
Video 1: Acid & Base INB pages, ChemEd X Vimeo Channel, 5/14/2023.
Questions
Concepts
Time Required
Preparation
Provide copies of the following for each student. Note that I have grouped the components by page. Please preview them before printing as some pages have multiple copies to minimize paper waste.
Spread 1:
Strong vs. Weak Acids & Bases,
Spread 2:
Measuring Acids & Bases, *A note on printing: If at all possible, I highly recommend you print the pH foldable in color. Additionally, if you need to limit your printing, the pH/pOH barndoor foldable is formatted to match the line spacing in college ruled composition books. There are two copies of the pH foldable per page, and students can line up the foldable to the lines on their page, eliminating the glue down, and you can have them fill in notes on equations directly on the notebook page.
Spread 3:
Attribution
Mihir Paranjape
Tom Jankowski
PhET Acid Base Solutions simulation (accessed 5/14/2023)