Soap Boat 2.0

Have you ever seen the soap boat experiment? Check out the video below.
The soap boat experiment makes use of the Marangoni Effect, the tendency of material to move from a region of low surface tension to a region of high surface tension. Because detergent has a lower surface tension than water, when a drop of detergent is added to the fuel reservoir in the boat, the surrounding water “pulls” the soap out of the reservoir. The flow of soap out of the reservoir provides thrust to push the boat forward.
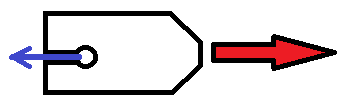
Figure 1 - Water, which has a higher surface tension than detergent, “pulls” detergent (blue arrow) out of the fuel reservoir in the soap boat. This flow of detergent propels the soap boat forward.
Unfortunately, the soap boat experiment works only once. That is, you can only add one drop of detergent to propel the boat forward; subsequent drops of detergent do not make the boat move. Because detergent is surfactant, detergent molecules added to water will spread over the surface of the water.
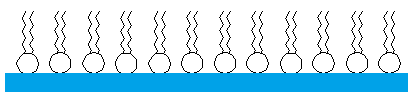
Figure 2 - Detergent molecules lining up on the surface of water
This lowers the surface tension of the water. Once detergent is added to the water, the now soapy water has the same surface tension as the detergent itself. Because the soapy water has the same surface tension as the detergent, the soapy water can’t pull additional drops of detergent out of the fuel reservoir. In order for the experiment to work again, you have to completely rinse the pan and boat free from residual detergent.
In the Journal of Chemical Education, Charles Renney, Ashley Brewer and Tiddo Mooibroek present an interesting twist on this classic experiment.1 In a sense, they have revolutionized the soap boat demonstration! Instead of adding detergent to power the soap boat, they add other low surface tension liquids like isopropyl alcohol or acetone. Although these liquids have low surface tension, they are not surfactants. When a non-surfactant, low surface tension liquid such as isopropyl alcohol is added to the fuel reservoir of the soap boat, the isopropyl alcohol is pulled out of the reservoir by water as described previously (Figure 1). However, because the isopropyl alcohol is not a surfactant, but rather is miscible with water, the alcohol molecules completely dissolve in the water. The alcohol molecules do not pile up on the surface of the water and therefore do not lower the surface tension of the water (Figure 3).
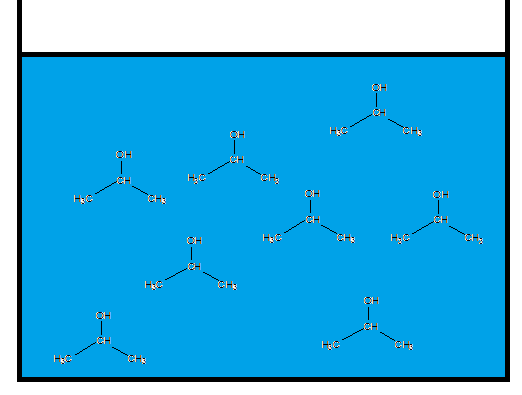
Figure 3 - Alcohol molecules do not spread out on the surface of water, but rather dissolve in the water.
Because alcohol molecules do not pile up on the surface of the water, the addition of alcohol to water has a negligible effect on the surface tension of water. Thus, water keeps its high surface tension when drops of alcohol are added to it, so drops of alcohol can be used multiple times to power a soap boat.
There are many possible variations on this experiment, some of which the authors discuss in their paper. For example, various dyes can be added to the low surface tension liquids to enable visualization of the Marangoni Effect in the soap boat experiment. The authors describe several boat designs, and test results from 15 different low-surface tension liquids they used to power Soap Boat 2.0. One interesting thing we found interesting is that glycerol (surface tension = 0.064 Nm-1), which has a surface tension very close to that of water (0.072 Nm-1) is a very poor fuel for the soap boat (as expected). Something fun we tried was testing soap boats in an old gutter. We wanted to see if our students could make a soap boat that traversed the entire length of the gutter.
I would love to hear some things that you learn as you try out this experiment, so feel free to comment!
Acknowledgement - I would like to thank Jin-Hwan No for helping me with many soap boat experiments.
1 - Easy Demonstration of the Marangoni Effect by Prolonged and Directional Motion: “Soap Boat 2.0”,C. Renney, A. Brewer, and T. J. Mooibroek,Journal of Chemical Education, 201390 (10), 1353-1357.
Comments
4Thanks, Tom!
So glad you decided to highlight this, Tom, or I might have missed it. The videos make it easy to get a quick taste of the experiment. I liked it so much that it seemed like a comment wasn't enough; I added a blog post about my experiences.
Share Your Thoughts