Increasing Access to Stoichiometry Through Differentiated In-Class Practice

In a recent post, I shared sample quiz questions as to how I have differentiated assessment within the mole unit. Here, I will share a specific multi-day sequence within the stoichiometry unit. I have written extensively about the project that drives this unit (within the following blog posts: Why consider trying project based learning?, Backwards planning your PBL unit - An Overview of an Entire Unit and What ARE my students actually learning during this long term project (PBL)?), but very little about specific learning tasks. Below is a two day sequence of stoichiometry practice that I set up in my classroom. Stations are set up around the room and students rotate as necessary.
Brief unit context (elaborated upon in the links above surrounding the project): The first day of the unit, students are introduced to the Pharmaceutical Challenge- can they make 2.00 g of an assigned nutrient for a patient requiring IV nutrition? They have no idea what stoichiometry is (yet), but this gets them literally asking me to teach it to them.
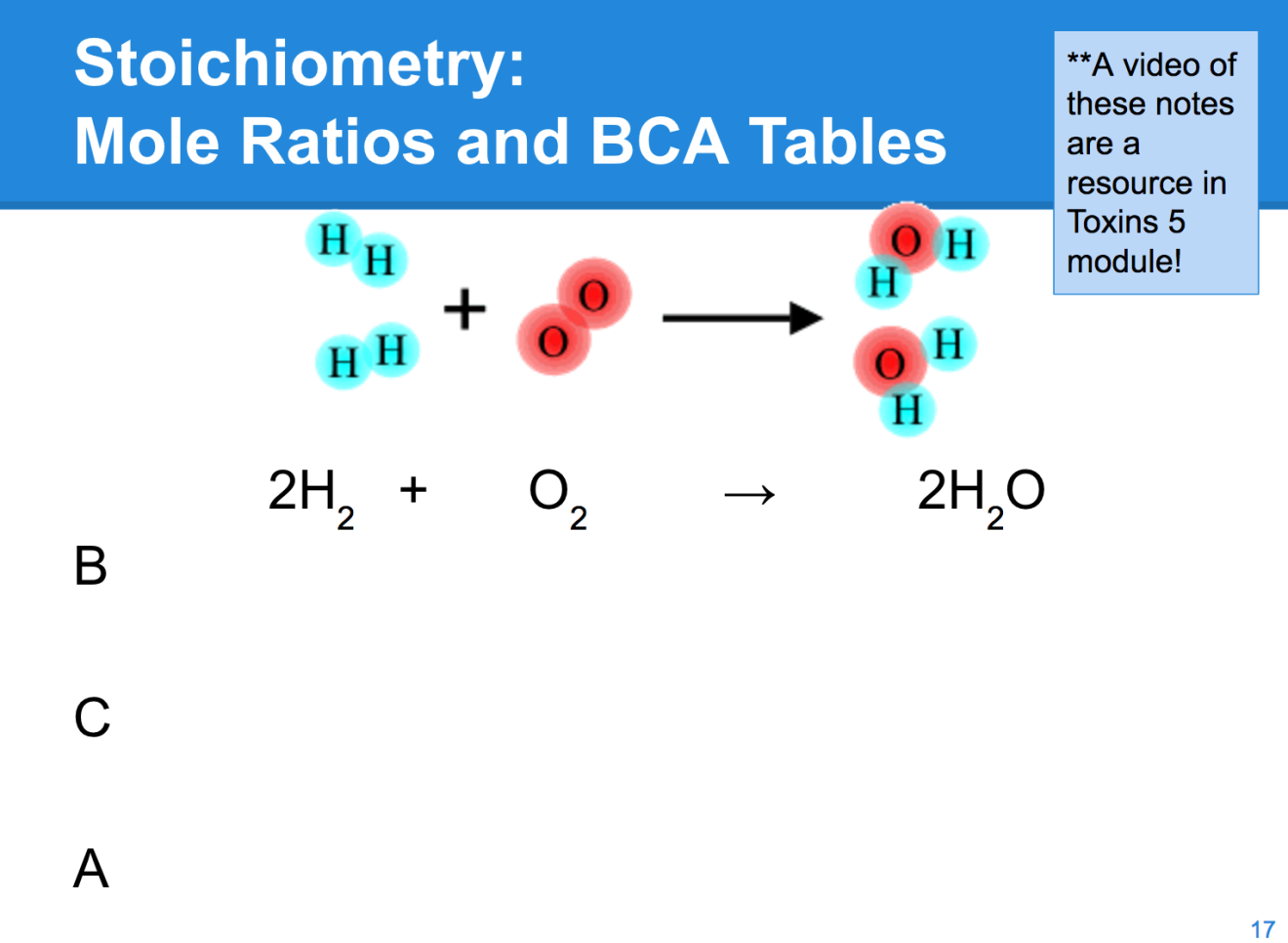
After my students experience the entry event, they complete the Flinn POGIL activity on mole ratios. This activity uses analogies to get students thinking about relationships between numbers of items and mass, which leads to a definition of the mole ratio and a few simple exercises with mole ratios and maybe a mass to mole to mole relationship. Most of my students got the big ideas, but over the years I realized most kids needed a bit more scaffolding to take the leap to independent practice. So after the POGIL, we wrap up with a short lecture and work a few more examples together. In this lecture, I introduce before, change, after (BCA) tables as a scaffold - I have found it to be a very nice bridge that allows even struggling students to do stoichiometric calculations (here is a great post from Lauren Stewart describing use of BCA). My top students benefit because this thinking will support them in more challenging AP chemistry work down the line. Depending on the class, I do 2-3 sample problems as shown below. (Note: I used to get frustrated that I'd invest all of this time for my students to do a POGIL and still need to lecture. Sometimes guided inquiry is nice, sometimes it isn't. I'm choosy. However, for stoichiometry, I used to only lecture and would have to do a million lecture examples and kids on the by and by did not demonstrate they understood what was going on, even if they could memorize the mathematical steps. With the POGIL, kids are way more in tune with what's going on even though I still need to lecture a bit.)


Then, students complete a mastery check (a self-graded mini-quiz to assess progress/mastery- this is mostly the provided POGIL check in with some tweaks).

From there, I propose to students as to how they may proceed to get 1.5 hours (1.5 class periods) of solid “drill and kill” stoichiometry practice. You may have found in your practice that some students after doing a POGIL do not need any other sample problems, while others can follow the guided lecture practice but struggle to apply on their own. For me, it has been emotionally painful to watch the brightest students breeze through independent practice and not get challenged while, at the same time, trying to support students who get the basic logic but cannot apply the logic on their own without some support. Thus, this differentiated practice was born and honed over the course of a few years. Note the big question of the project, as well as these instructions, stay on the board the whole class period- even though I don’t make a big deal about the project, it is a reminder in the background that this is the purpose of all of the practice.

There are five levels of practice. I push down the urge to micromanage this next part- student determine where to start. I give them a rough guide to use their mastery check- 100% or just small silly mistake? Start at level 3 or 4. Totally lost on the mastery check? Level 1. Got the basic idea but didn’t finish the mastery check? Start at level 2 or 3. However, this is a squishy description for me and I used to stress about students landing in the “right” place. Happily, for the most part, my agony was unfounded and students are, for the most part, challenged. [Interesting side-note/over-generalization: sometimes ladies choose a “level” lower than I think they should be, and boys choose a level “higher” than I would have assessed… not everyone, but some interesting cases.] Sometimes, if a student struggles for a LONG time, I might suggest moving down and then coming back to try again after a little more practice. It has never been a huge deal to ask a student to move.
As you peruse the practice problems, you will notice that level 1 is simply mole to mole practice. In level 2, I add grams to mole and then mole to mole. Level 3 incorporates more units. Levels 4 and 5 are just fun problems that use mole ratios but require a bit more creativity and extrapolation of the basic ideas to solve.
My eight lab tables are roughly one-two for levels 1,4, and 5 and two- three for levels 2 and 3. There are labels and many copies of the corresponding practice set in a folder at the table. Here is a representative table label:

When do students move to the next level? Students must check their work in a different color before checking in with me to record their progress in a spreadsheet. I can then ask questions about their annotations and use of BCA tables and such as I see fit.
What students see online is one succinct document shown below. Even though I print the blank practice problems, posting the originals is useful for students who are absent or want to work more at home (which does happen!).

There is a whole-class incentive in addition to a personal incentive of a completion quiz grade (note: this is the ONLY completion quiz I ever use- that is the extent of my bribery to do a good job and it really works for the most part). The class chooses the incentive they work toward- the example class below chose for a few bonus points on their unit test, while other classes asked for bonus points on a project presentation or to drop the lowest quiz grade (for that last one, I upped the number of points needed as a class). Most classes meet the goal even if there are 1 or 2 absent students.

What am I doing as students work? I am pacing the room, checking in individually with students or small groups of students for prodding, and just trying to encourage them to make progress and to keep pushing forward.
Finally, at the end of this practice work time, I force my students to take 15 minutes or so to gather their thoughts surrounding how this applies to their project at hand. The goal for this time isn’t so much to DO the math for their project than to summarize WHAT they will need to do.

As I reiterate in almost every post, it has taken me years to build the flow and find problems that fit my students. This is a culmination of many things: my novice attempts at teaching with BCA tables, figuring out trouble points in their big old stoichiometry project, and more. Students still have an un-differentiated practice set before the unit exam, because in my mind, this still isn’t enough practice for all, but it’s a good start. Feel free to use this and try it in your classroom, or use this as inspiration to give your materials a remix. Thank you as always for your readership- it’s an honor to write for ChemEdX.
Materials
Worksheets from supporting information set up at stations.
Background
Procedure
After an introduction to mole ratios and before, change, after tables, students engage in differentitated stoichiometry practice problems as outlined above.
Questions
Concepts
Mole ratios; unit conversions
Time Required
POGIL: ~1 hour
Lecture: ~30 minutes
Mastery check, differentiated practice, project planning: ~2 hours
Preparation
Print out practice problems, post answers, print table labels (I like to use different colored paper for each document).
Attribution
Source for many of the problems: https://chemfiesta.org/2014/10/31/worksheets/; other problems are from miscellaneous textbook problems and such- for instance, Level 5 is mostly “intro” AP chemistry stoichiometry and one problem from "Solving Real Problems in Chemistry" from Pacific Crest. Activities are posted below. If I missed you in attribution (as this has been compiled over quite some time), please let me know and I will give you credit!
Comments
5Holey Moley!
Hi,
I am so glad that I stumbled upon your activity. I have been struggling with this... yesterday was a lesson in frustration, as we were tackling limiting reagents... and some students "get it" and fly through the problems, while I have a small core group who are still struggling to determine moles... I plan to try this next year!
SAM
Best wishes
Hi SAM-
Thanks for joining the conversation (and the pun)!
I truly hope that this is useful for you, and that you make it even better (and share how you do so)!
Share Your Thoughts