Quick Lab on Mass vs Moles

This year, I tried something different with my course progression - instead of doing chemical quantities first and teaching students to convert from grams to mole and then move into stoichiometry, I began with mole to mole stoichiometry and BCA tables. My hope was students would have an understanding of the mole and its purpose in chemistry, giving relevance to quantities. After a week or two of working with BCA tables using only moles, students were able to identify limiting and excess reactants and determine the number of moles of reactants needed and products produced in reactions when given a molar amount in the prompt. After assessing this part, we paused BCA tables to talk about unit conversions. We discussed the fact that moles cannot be measured directly in the lab, and so we measure them indirectly. We then spent some time practicing those conversions and we quizzed mole conversions (it went really well). So far, I like this change much better than what we had previously done. Students see the purpose in mole conversions - we use grams and liters to measure moles.
When we were ready to come back to BCA tables, I wanted to remind students of the connections between mass and moles in terms of reactions and to prove a point: mole conversions are necessary. To drive this point home, my colleague Tom Jankowski and I designed a quick lab.
Students measure out equal amounts of three metals: magnesium, iron and tin. They add the metals samples to a solution of copper (II) chloride and make observations about the reaction (see figure 1). I created a placemat lab handout that contained everything they needed to do. It includes a diagram outlining the procedure, and students were asked to think about the results both qualitatively and quantitatively. By using a copper solution, the color of the products could be used to support thoughts of what was limiting or excess in each situation.

Figure 1. Sample student observations.
The lab itself only takes about 10 minutes to perform. We did it on a small scale, using 0.25 grams of each metal and 5 mL of 1.0 M CuCl2. The metals were chosen so that one would be the limiting reagent (by roughly half), one would be similar to the number of moles of Cu2+, and the other would be in excess (by roughly twice). By choosing this range, small variations in measurement should not change the end result. Waste was easy to dispose of, and the lab itself is safe but engaging.

Figure 2. CER portion of worksheet with sample student answers.
The final analysis asked students to respond to a CER to think about what mattered more: mass or moles (see figure 2). Overall, it worked very well. Students were able to see that the moles are what impacted the reactions, as the reactions were visibly different and proceeded to different extents. We had them analyze using all points of Johnstone’s Triangle, and students in fact discussed all three points in their analyses (see figure 3). In addition, we have been reminding students of the results from this lab when using BCA tables. A common error for students with BCA tables is to use units other than moles in the table. After this lab, I have fewer students than before making this mistake, and when they do initially write their grams in the table, they are quick to catch their error before I even have to point it out.
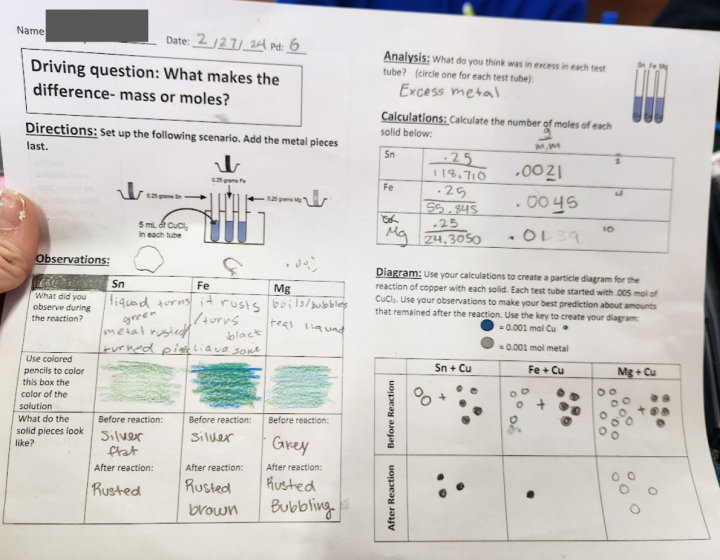
Figure 3. Full placemat lab handout with sample student answers.
This quick lab will definitely be a keeper for us moving forward. We will make a few tweaks. The biggest change will be to have all three metals in the same form. The samples we had on hand were all different - iron wire, magnesium ribbon and tin foil. Next year we’ll try to procure sheets of iron and tin, making the results more similar and increasing the surface area to allow a more complete reaction in a short amount of time. While we feel we can make it even better, the student take-aways are already solid and enduring so it was a win!
I've shared the placemat handout in Supporting Information so you can use this activity with your students. I love that it really helped students to have a very visual reason for why we convert mass into moles before we perform stoichiometry calculations. It's also a low-prep (quick) setup that gives your students another lab experience, and the more we can do that, the better our students will be at tying their in-class work to real world chemical phenomena. If you try it out, I hope you will let me know how it goes for your students!