Lesson to determine the relationships between acid strength, Ka, pH, Molarity, and percent ionization

After teaching the concepts and calculations for acid and base strength, concentration, percent ionization, and pH I noticed many of my students were struggling to make meaningful connections between these calculations. To excel in the topic of acid and base chemistry, students must be able to make the connection that the pH is a value determined by the number of hydronium ions present in a solution. The number of hydronium ions present is dependent on both the Molarity of the solution and the strength of the solute (the percent the acid or base will ionize).
Proficient students should be able to calculate the pH, Molarity, percent ionization, and Ka of an acid or base, as well as create and evaluate particle diagram models depicting various acidic and basic solutions, construct and evaluate graphs and trends of this data, and analyze and interpret the patterns and trends in lab data. Proficient students should also be able to use these skills to make predictions and arguments about additional solutions to explain chemical phenomena. This lesson was created to strengthen the understanding of the relationships between these concepts and skills.
Many students tend to associate pH values only with the strength of the acid. The major misconception is that acids with pH values approximately less than 2 are strong and acids with pH values of 3-7 are weak. This can be a fatal mistake, as the same student may think they can ingest all weak acids, like citric acid or vinegar, regardless of their concentration. Before this lesson, the concept of acids and bases was introduced with this misconception in mind. Students had to create models depicting many different concentrations of various weak and strong acids and bases. As lessons continued, my lessons had a strong focus on mathematical computations, required by the Advanced Placement Exam. This lesson was designed to refocus the instruction to the particulate, macroscopic, and symbolic representations of their calculations.
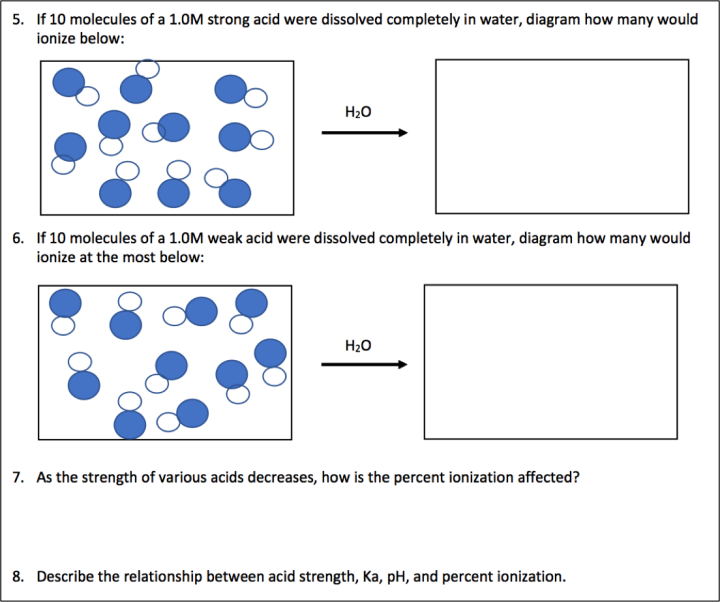
Figure 1: A portion of the lesson worksheet found in the Supporting Information.
Students are asked to calculate the pH and percent ionization of 1M solutions of varying acid strength (given by Ka value) (see Figure 1). These calculations are then analyzed and interpreted to determine relationships between Ka and pH graphically, in a particulate model, and narratively. Then students are asked to calculate the pH and percent ionization of one weak acid at varying concentrations to identify the relationship between the two factors graphically, in particulate diagrams, and narratively. Finally students are asked to explain an interesting phenomenon associated with percent ionization of weak acids as they are diluted. This work allows students to see the concept from multiple perspectives in order to draw a complete correlation between pH, acid strength, and Molarity.
Following this lesson, students are required to use their understanding of pH calculations and weak acids and bases in order to identify and calculate the pH of solutions at various points during a titration. Additionally, students have to identify and calculate the pH of buffer systems created with weak acids and bases with their conjugate salts. Without a firm understanding of the solutions at the particulate level, students will easily be confused and overwhelmed by the calculations for titrations and buffers and many struggling learners resort to using systematic calculations which will fail them in more complex and abstract problems that arise. Specifically, students will be challenged with questions asking how errors in their labs may affect calculated Molarities, molar masses, and pH values. Error analysis questions are probably the most challenging questions for students in the lab, rigorously assessing how well the student understands the concept beyond simple calculations. To clarify these concepts we need a solid understanding of the composition and reactivity of the initial acids and bases used in titration and buffers. Students leaving AP Chemistry will also need to be able to apply this knowledge to AP Biology topics and college level classes if this is their major. But more importantly, all students can benefit from an understanding of pH and acid base chemistry. Though a non-chemist may never complete a titration again in their life, per se, they may have to clean up an acid spill, settle an upset stomach, or regulate the pH of a pool or fish tank. Acids and bases are part of our day to day lives and adding acids and bases to our household solutions to neutralize them is fairly common. Additionally, systems like pools, fish tanks, contact lens solutions, and many others serve as ordinary buffer systems. Understanding how the buffer system resists changes in pH can help students be better global citizens.
Due to the socioeconomic status of our district, our students do not all have home computers or consistent internet access. Computer simulations and online demonstrations such as Sunflower Learning and phET interactives are great tools to model how our macroscopic world interacts at the particulate level. If I were to complete the lesson again in a standard class setting I would have students test various solution noted in the activity for their pH as evidence for their work. Adding in testing of solutions would help tie in the macroscopic observations with their particulate diagrams, graphs, and calculations for a well-rounded lesson.
To formatively assess this lesson I made sure to move around the room and check on student work as it was being competed. I spoke to students directly when I was able to identify a misconception or noticed a student was struggling to answer a problem. If a student was struggling, I asked questions to elicit their prior knowledge and build connections to the question being addressed. To close the lesson, students were given four questions similar to the activity but in terms of basic solutions (our activity was acids only). The students were asked to individually calculate, draw particulate diagrams, describe relationships, and sketch graphs about the pH of bases, their strength, percent ionization, and Kb values (included in supplemental materials). If students could translate the work we completed as a class to another situation in four different ways (mathematically, graphically, diagrammatically, and narratively), it would prove to me that they were capable at this level of acid base chemistry to move on.