Exploring The “Two-Faced” Thionin Reaction
The chemical reaction between violet-colored thionin and iron (II) ions produces a leucothionin, a colorless compound (Equation 1).1,2
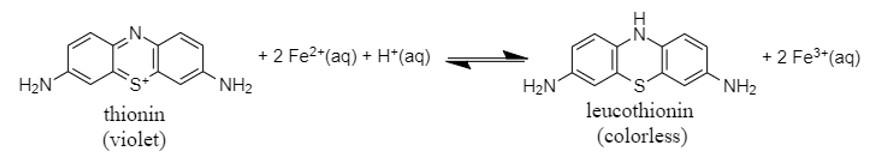
Equation 1
Because DGo = + 75 kJ mol-1 for this reaction (see below), it is not spontaneous. However, light energy can be used to power the reaction. Thus, the purple color of a solution of thionin and iron (II) ions is observed to fade when exposed to light. When the light is turned off, the color returns as the reaction proceeds spontaneously in the reverse direction. These facts allow for the setup of some fascinating chemical reactions (Video 1).3
Video 1: Chemistry lights up my life, @pchemstud on TikTok, accessed April 18, 2022.
The absorption spectrum of thionin indicates that it absorbs yellow light very well (Figure 1).
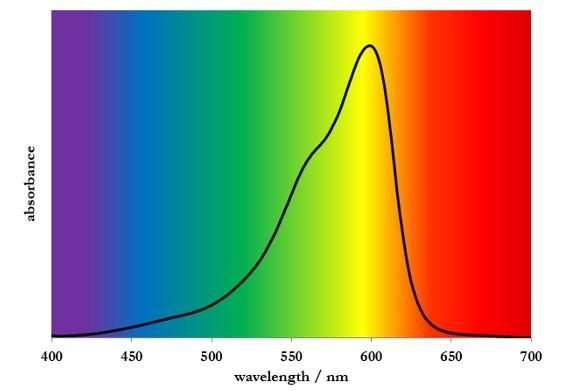
Figure 1: Absorption spectrum of thionin
Because of this, I wondered if it could be demonstrated that yellow light drives this reaction (Equation 1). Check out the experiments I did to test for this (Video 2).4
Video 2: A Chemical Reaction that is Powered by Light, Tommy Technetium YouTube Channel, April 10, 2022
Connections to the chemistry curriculum
This experiment may be used to connect to topics in electrochemistry, thermodynamics, and quantum chemistry.
Connections to electrochemistry
The overall reaction (Equation 1) involves the oxidation of Fe2+ and the reduction of thionin. Therefore, we analyze Equation 1 by considering the following half reactions:
thionin + H+(aq) + 2e- → leucothionin Eo = 0.38V Equation 2
Fe3+(aq) + e- → Fe2+(aq) Eo = +0.77V Equation 3
Notice that Equation 1 results if we subtract twice Equation 3 from Equation 2. The overall cell potential (Eocell = Eored – Eoox) for Equation 1 is therefore -0.39V. This negative cell potential indicates that the reaction should be non-spontaneous, in accordance with the observation that the reaction requires light energy to proceed.
Connections to thermodynamics
From the calculated cell potential, the change in the standard Gibbs energy (DGo) can be calculated using:
DGo = - nFEo Equation 4
Where n is the number of electrons transferred in the reaction, F is Faraday’s constant (96485 C mol-1), and Eo is the cell potential for the reaction described in Equation 1. For Equation 1, n = 2 and Eo = -0.39 V. Substitutions of these values into Equation 4 yields +75 kJ mol-1. Once again, the positive Gibbs energy for this reaction is consistent with the non-spontaneity of Equation 1, and the fact that light energy is required to drive the formation of leucothionin from thionin and iron (II) ions. On the other hand, DGo = -75 kJ mol-1 for the reverse reaction (Equation 1). Therefore, the color returns spontaneously when the light is turned off.
Connection to quantum chemistry
As indicated in Video 2, thionin absorbs yellow light quite well. Indeed, the absorption spectrum of thionin peaks at about 600 nm (Figure 1). The energy, E, of a yellow photon can be calculated using:
E = hc/l Equation 5
Where h is Planck’s constant (6.626 x 10-34 J s), c is the speed of light in a vacuum (3.0 x 108 m s-1), and l is the wavelength of the photon in question. Substitution of these constants and l = 600 x 10-9 m into Equation 5 yields 3.31 x 10-19 J for a yellow photon. Multiplication of this energy by 6.02 x 1023 photons per mole means that 200 kJ of energy accompanies every mole of yellow photons. The energy per mole of yellow photons clearly exceeds the 75 kJ mol-1 necessary to drive Equation 1 to the right. This is consistent with the observation that yellow light absorbed by the thionin reaction mixture caused the thionin to fade (Video 2).
Conclusion
The reaction between thionin and iron (II) is very easy to set up, connects seamlessly to a variety of chemical topics, and is a real crowd pleaser. It is definitely worth performing for your students. Drop me a line in the comments if you try out this experiment. If you have already conducted this experiment, let me know how you include it in your chemistry classes.
Happy Experimenting!
References
1. Thionin – The Two Faced Solution, Flinn Scientific, Inc., 2017. https://www.flinnsci.com/api/library/Download/1e347e52d1fa4253b324d388aea826d1 (accessed April, 2022).
2. Shakhashiri, B. Z. Chemical Demonstrations: A Handbook for Teachers of Chemistry; University of Wisconsin Press: 2011; Vol. 5, pp 249−255.
3. Chemistry lights up my life, Tommy Technetium, TikTok video. https://www.tiktok.com/@pchemstud/video/7083104858265488686 (accessed April, 2022).
4. A Chemical Reaction that is Powered by Light, Tommy Technetium, YouTube Video. https://www.youtube.com/watch?v=j_ZnD71zbSM (accessed April, 2022).