Chemistry of Canning: Classroom Activity Dealing with Concentrations and Molecular Structure

First, thanks for reading! Second, share liberally and share often. I hope you and your students enjoy and find useful this classroom activity dealing, in part, with the canning of fruits. My motivation in designing this activity is that I like to can food, especially fruits like yellow or white peaches (which are in season), for long-term storage and delectable eating months later. And I hope students find an interest in possibly learning to do the same. To my mostly insensitive tastebuds, home-canned fruits (and other foods in general) taste better than the same fruit in metal cans found in grocery stores.
The two main chemistry topics emphasized in the questions (Qs) below deal with concentration and molecular structure. The concentration Qs are algorithmic in type whereas the structure Qs are qualitative and/or conceptual. The focus is on two unsaturated organic molecules- citric and ascorbic acid. Qs 1-5 are applicable to both Gen Chem 1 and OChem 1, with modifications where deemed appropriate by the instructor. Q6 is (likely) exclusive to OChem 1. Below some of the Qs is commentary about the Q. Qs are separated by a horizontal line for visual clarity. I recommend using the Think-Pair-Share instructional approach for this classroom activity, either individually or in groups. In general, with other classroom activities like this one, I find- based on my experience- that a group of 2 or 3 students is optimal for authentic intellectual engagement with minimal distractions such as irrelevant side conversations.
A confession: In Gen Chem 1 I teach molecular structure from an organic chemistry perspective. In doing so I probably am committing chemistry education heresy in not really focusing (too much) on VSEPR. I have found that students are most interested in organic molecules. And with organic molecular model kits and a protractor in hand, it is just easier for students to 'get' 2- and 3-D structure relationships and approximate bond angles. It also allows for discussion about the limitations of models. And it is engaging to build models.
Some introductory comments: Light colored fruits such as apples, peaches, nectarines, apricots, and bananas darken when cut and then exposed to air. Specifically, the molecular oxygen gas in air, coupled with oxidase enzymes and polyphenols in the fruit, combine in oxidatively-driven polymerization reactions to produce browning. In general, to Americans browned fruit is not visually appealing and this (may) prevent(s) consumption of the browned fruit because it is erroneously believed the fruit has gone bad or is rotten. (Of course, taken too far and with time the food does rot but that is a different chemistry going on). In canning light colored fruits, a chemical pretreatment is used to prevent darkening prior to canning (or dehydrating). The questions center around two common organic chemicals used to prevent fruit browning during canning. The real name/identity of the two molecules is never revealed to students, only the structures.
Q1: The two organic chemicals used to prevent browning in light colored fruit are shown below. The percent by mass composition for one is 40.9% C. Based on this information alone, which molecule below- the one on the right, or the left- has 40.9% C by mass? To support your claim you need to show the correct calculations with correct units. When done, find another student or group and compare/contrast answers. Reconcile any differences in your answers.

For instructor: Obviously not all the hydrogen atoms are shown for either molecule. And nor are the carbon atoms explicitly identified with the element symbol C. By the time I have done this activity (or a related one) students know that carbon has four covalent bonds, hydrogen one, and oxygen two. And that each vertice, where a nminimum of two solid lines (bonds) meet, represents a carbon atom. Students then should deduce that where two solid lines meet that site represents carbon and that for two lines there are two hydrogen atoms each singly bonded to the carbon but are not shown. Where three solid lines meet that vertice represents one C-H bond, not shown.
Q2: Which choice(s) below, if any, is (are) true for the two molecules above in Q1? MM = molar mass When done, find another student or group and compare/contrast answers. Reconcile any differences in your answers.
Q3: When canning white or yellow peaches, I use 3000mg of the molecule below to make an aqueous preservative solution. To do so the 3000mg is dissolved in one gallon of water. I then place the peeled and cut peaches in the aqueous preservation solution for a few minutes or so to prevent against future browning. What then is the concentration of the preservative solution in molarity? When done, find another student or group and compare/contrast your molarity value. Reconcile any differences.

For instructor: I expect students to figure out MM from the molecular structure. And I do not give the conversion constant between gallons and liters (needed for molarity). Why not do this? Either they know it already (unlikely for most), or they need to find it. I am a proponent of not giving all the info needed to find an answer. Rather students need to find the relevant info based on what the Q asks. Why do I believe this? It mimics reality in our everyday lives.
Q4: Suppose instead of using the molecule below left (molecule Y) to make the aqueous preservative solution as in Q3, the other organic molecule below right is used. The same volume of preservation solution in Q3 is made with Z. And the same molarity of preservation solution is made as in Q3 but this time with molecule Z. Based on this information, which choice(s), if any, is (are) true in making the preservation solution instead with molecule Z? Choice (9): none of the choices. When done, find another student or group and compare/contrast answers. Reconcile any differences in your answers.

Q5: Which choice(s) below, if any, is (are) true with respect to molecules Y and Z? When done, find another student or group and compare/contrast answers. Reconcile any differences in your answers.
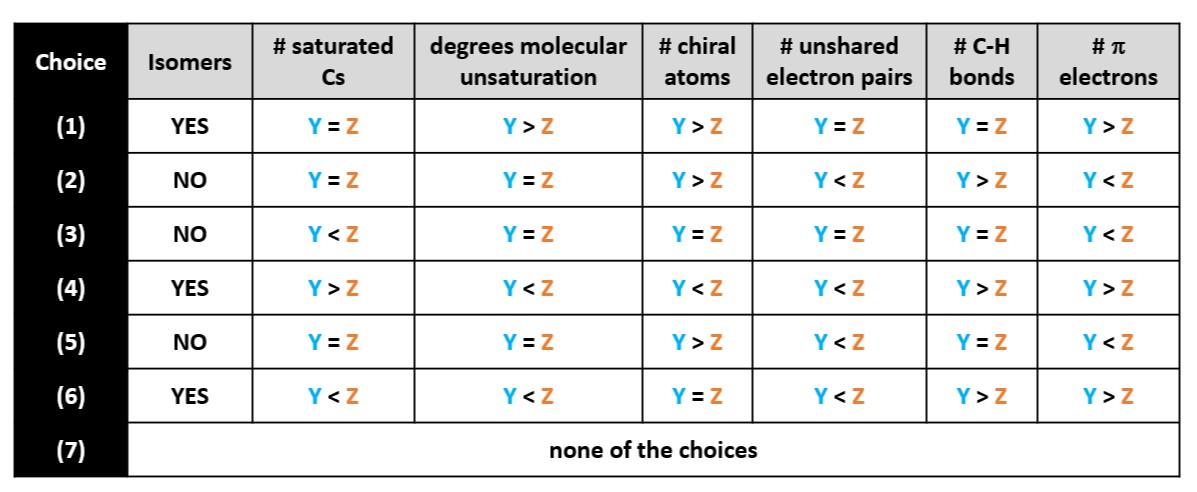
For instructor: Below is an alternative format for choices to those above. The choices below may be more amendable to Gen Chem 1 if isomers is not taught as part of molecular structure. Also, column 4 which deals with individual chiral atoms may not be applicable. It is included as a possible choice.
Q6: Which choice(s) below, if any, is (are) true with respect to molecules Y and Z? When done, find another student or group and compare/contrast answers. Reconcile any differences in your answers.

For instructor: Q6 is definitely for OChem 1 students.
In closing, interested in the historical timeline of food canning? Check out the website below.

Cheerio,
SJD