 This inventory consists of 22 multiple choice questions. Carefully consider each question and indicate the one best answer for each. Several of the questions are paired. In these cases, the first question asks about a chemical or physical effect. The second question then asks for the reason for the observed effect. This inventory consists of 22 multiple choice questions. Carefully consider each question and indicate the one best answer for each. Several of the questions are paired. In these cases, the first question asks about a chemical or physical effect. The second question then asks for the reason for the observed effect.
1. Which of the following must be the same before and after a chemical reaction?
- The sum of the masses of all substances involved.
- The number of molecules of all substances involved.
- The number of atoms of each type involved.
- Both (a) and (c) must be the same.
- (e) Each of the answers (a), (b), and (c) must be the same.
2. Assume a beaker of pure water has been boiling for 30 minutes. What is in the bubbles in the boiling water?
- Air.
- Oxygen gas and hydrogen gas.
- Oxygen.
- Water vapor.
- Heat.
3. A glass of cold milk sometimes forms a coat of water on the outside of the glass (Often referred to as 'sweat'). How does most of the water get there?
- Water evaporates from the milk and condenses on the outside of the glass.
- The glass acts like a semi-permeable membrane and allows the water to pass, but not the milk.
- Water vapor condenses from the air.
- The coldness causes oxygen and hydrogen from the air combine on the glass forming water.
4. What is the mass of the solution when 1 pound of salt is dissolved in 20 pounds of water?
- 19 Pounds.
- 20 Pounds.
- Between 20 and 21 pounds.
- 21 pounds.
- More than 21 pounds.
5. The diagram represents a mixture of S atoms and O2 molecules in a closed container.

Which diagram shows the results after the mixture reacts as completely as possible according to the equation:
2S + 3O2 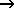 2SO3 2SO3

6. The circle on the left shows a magnified view of a very small portion of liquid water in a closed container.

What would the magnified view show after the water evaporates?

7. True or False? When a match burns, some matter is destroyed.
- True
- False
8. What is the reason for your answer to question 7?
- This chemical reaction destroys matter.
- Matter is consumed by the flame.
- The mass of ash is less than the match it came from.
- The atoms are not destroyed, they are only rearranged.
- The match weighs less after burning.
9. Heat is given off when hydrogen burns in air according to the equation 2H2 + O2 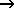 2H2O 2H2O
Which of the following is responsible for the heat?
- Breaking hydrogen bonds gives off energy.
- Breaking oxygen bonds gives off energy.
- Forming hydrogen-oxygen bonds gives off energy.
- Both (a) and (b) are responsible.
- (a), (b), and (c) are responsible.
10. Two ice cubes are floating in water: 
After the ice melts, will the water level be:
- higher?
- lower?
- the same?
11. What is the reason for your answer to question 10?
- The weight of water displaced is equal to the weight of the ice.
- Water is more dense in its solid form (ice).
- Water molecules displace more volume than ice molecules.
- The water from the ice melting changes the water level.
- When ice melts, its molecules expand.
12. A 1.0-gram sample of solid iodine is placed in a tube and the tube is sealed after all of the air is removed. The tube and the solid iodine together weigh 27.0 grams. 
The tube is then heated until all of the iodine evaporates and the tube is filled with iodine gas. Will the weight after heating be:
- less than 26.0 grams.
- 26.0 grams.
- 27.0 grams.
- 28.0 grams.
- more than 28.0 grams.
13. What is the reason for your answer to question 12?
- A gas weighs less than a solid.
- Mass is conserved.
- Iodine gas is less dense than solid iodine.
- Gasses rise.
- Iodine gas is lighter than air.
14. What is the approximate number of carbon atoms it would take placed next to each other to make a line that would cross this dot: 
- 4
- 200
- 30,000,000
- 6.02 x 1023
15. Figure 1 represents a 1.0 L solution of sugar dissolved in water. The dots in the magnification circle represent the sugar molecules. In order to simplify the diagram, the water molecules have not been shown. 
Figure 1
Which response represents the view after 1.0 L of water was added (Figure 2).

Figure 2
16. 100 mL of water at 25°C and 100 mL of alcohol at 25°C are both heated at the same rate under identical conditions. After 3 minutes the temperature of the alcohol is 50°C. Two minutes later the temperature of the water is 50°C. Which liquid received more heat as it warmed to 50°C?
- The water.
- The alcohol.
- Both received the same amount of heat.
- It is impossible to tell from the information given.
17. What is the reason for your answer to question 16?
- Water has a higher boiling point then the alcohol.
- Water takes longer to change its temperature than the alcohol.
- Both increased their temperatures 25°C.
- Alcohol has a lower density and vapor pressure.
- Alcohol has a higher specific heat so it heats faster.
18. Iron combines with oxygen and water from the air to form rust. If an iron nail were allowed to rust completely, one should find that the rust weighs:
- less than the nail it came from.
- the same as the nail it came from.
- more than the nail it came from.
- It is impossible to predict.
19. What is the reason for your answer to question 18?
- Rusting makes the nail lighter.
- Rust contains iron and oxygen.
- The nail flakes away.
- The iron from the nail is destroyed.
- The flaky rust weighs less than iron.
20. Salt is added to water and the mixture is stirred until no more salt dissolves. The salt that does not dissolve is allowed to settle out. What happens to the concentration of salt in solution if water evaporates until the volume of the solution is half the original volume? (Assume temperature remains constant.) 
The concentration
- increases.
- decreases.
- stays the same.
21. What is the reason for your answer to question 20?
- There is the same amount of salt in less water.
- More solid salt forms.
- Salt does not evaporate and is left in solution.
- There is less water.
22. Following is a list of properties of a sample of solid sulfur:
- Brittle, crystalline solid.
- Melting point of 113oC.
- Density of 2.1 g/cm3.
- Combines with oxygen to form sulfur dioxide
Which, if any, of these properties would be the same for one single atom of sulfur obtained from the sample?
- i and ii only.
- iii and iv only.
- iv only.
- All of these properties would be the same.
- None of these properties would be the same.
|



